Main Trial
DoubleD-Trial is an academic, investigator-initiated, open-label randomised study. The aim is to evaluate whether early double sequential defibrillation (DSD) improves survival compared with standard defibrillation in out-of-hospital cardiac arrest (OHCA) patients with an initial shockable rhythm who remain in cardiac arrest after at least one standard shock. The trial is coordinated by the Centre for Resuscitation Science, Karolinska Institutet (sponsor).
Inclusion is performed by EMS units with two study defibrillators available (currently Corpuls3). Eligible patients are over 18 years old with VT/VF after at least one standard defibrillation with pads in antero-lateral position.
A pilot study (n=40) in West Sweden was completed in April 2025. The main trial started recruitment on 8 September 2025.
Inclusion Criteria
✅ OHCA with an initial shockable rhythm and at least one defibrillation delivered in the standard A-L position
✅Two study-specific defibrillators available on scene
Exclusion Criteria
❌ Age: under 18 years
❌ Known or obvious pregnancy
❌ Known pre-existing Do Not Attempt Resuscitation order
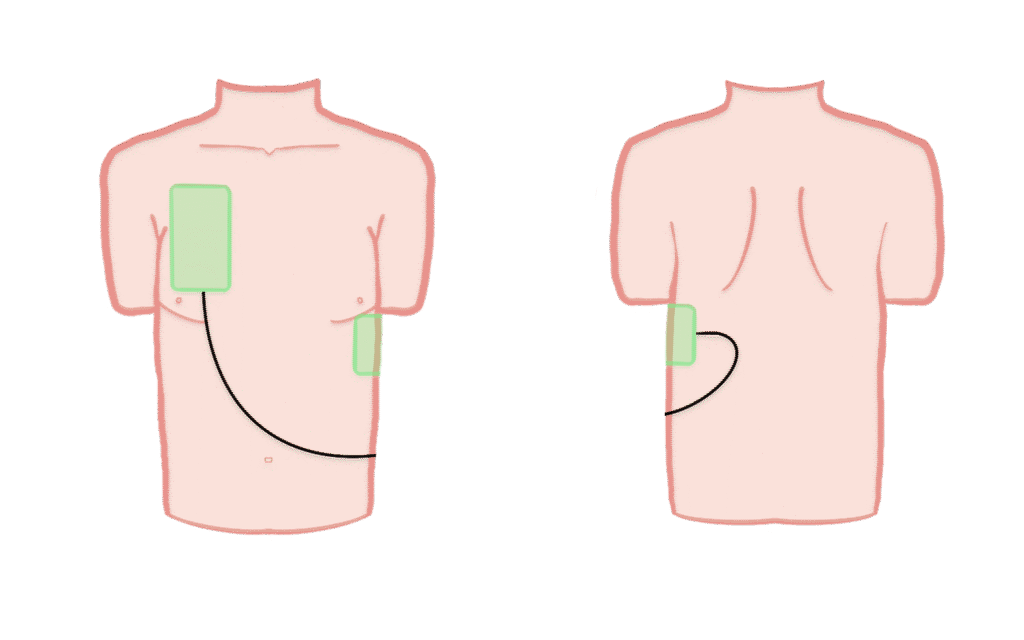
Control group
Patients in the control group receive standard Advanced Life Support. Defibrillation is performed with one defibrillator in the standard antero-lateral position and continued until return of spontaneous circulation, termination of resuscitation, or arrival at hospital.
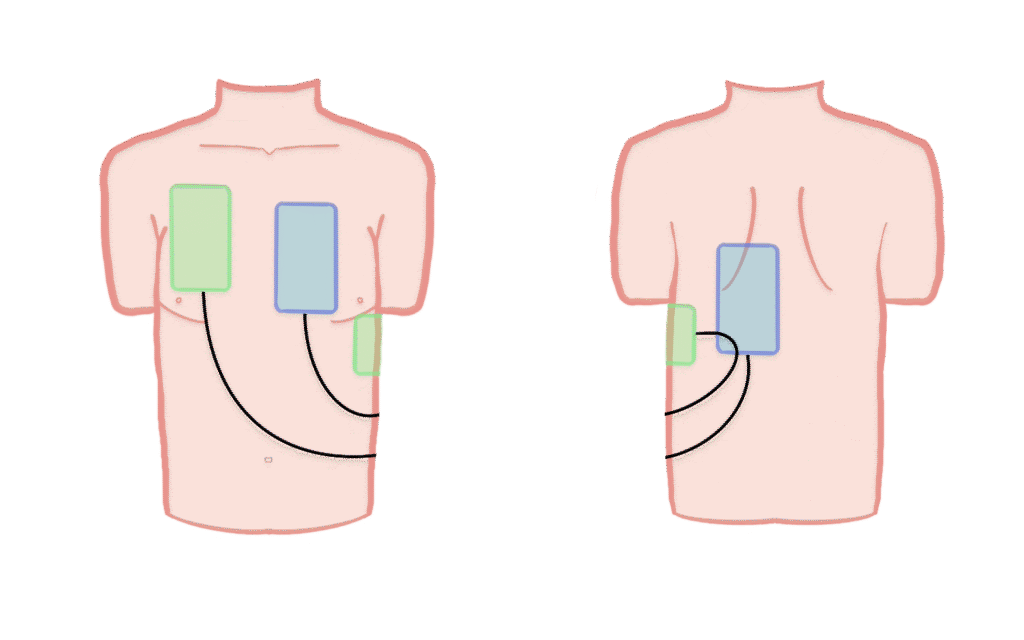
Intervention group
Patients randomised to the intervention group will receive Double Sequential Defibrillation (DSD). A second defibrillator is applied with pads in the antero-posterior position, and if VT/VF persists, shocks are delivered sequentially. All further defibrillations follow the DSD strategy until ROSC, termination of resuscitation, or transport to hospital.
Flowchart

